Helping Pharma IT to leverage technology and elevate clinical development business.
Clinical Processes
Our experience spans end-to-end Clinical Development processes and we can assist you in redefining your processes to derive the most out of today’s technologies.
Clinical IT
With deep industry knowledge, coupled with extensive technology expertise, we can support the evaluation, design, implementation and adoption of new IT technologies within your organization.
Clinical Data
We have rich clinical data domain expertise through decades of working with leading pharmaceutical, enabling you to gain the best from your clinical data.
About Us
Tomorrow’s medicines cannot be developed with yesterday’s processes. That is why we established intilaris.
intilaris LifeSciences, a Clinical Development Consulting, company optimizes Clinical Development and Operations through standards and data driven processes.

Clinical solutions
Learn more about us

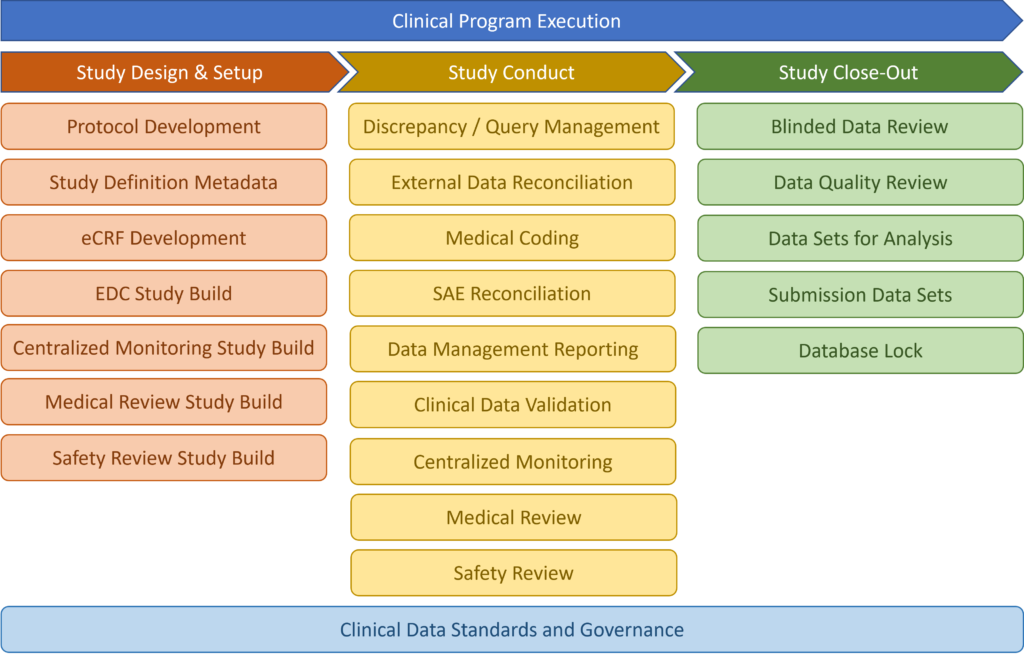
Our scope
We specialize in processes, IT solutions and clinical data flow within clinical program execution space: from study design and setup, conduct to study close-out.
Process analysis and optimization
We help our clients by facilitating the development of optimized To-Be processes from the As-Is state and a gap analysis to improve their clinical data flow and enable clinical trial digitalization. Our consultants provide unbiased input and facilitate the process optimization across organizational departments.
What can our clients expect from us

Responsibility
We will take responsibility, your problems become our problems and we will work hard on providing you with an appropriate solution.

Expertise
We are pharma experts, and will know how to solve your issues using experiences from the industry peers.

Trust
We will be honest and challenging. Our opinions are independent and provider agnostic. We always tell you how it is.

Torsten Friedel
Director Consulting
We assist Senior Management to elevate insights and experiences by systematically addressing the ways to create added value across the organization.
We support Life Sciences and Pharma customers in the R&D Digital Transformation and Agile GxP Systems Implementation.

Dr. Djenan Ganic
Director Business Solutions
Latest news
Our latest blog posts
What our clients say
Let's talk about your next project
Combine our knowledge and expertise – Better Together. Let’s Talk.









